Researchers scale up tiny actuator inspired by muscle
Researchers at CU Boulder are collaborating to develop a new kind of biocompatible actuator that contracts and relaxes in only one dimension, like muscles. Their research may one day enable soft machines to fully integrate with our bodies to deliver drugs, target tumors, or repair aging or dysfunctional tissue.

Assistant Professor Carson Bruns (left) and Professor Franck Vernerey (right).
Professor Franck Vernerey of the Paul M. Rady Department of Mechanical Engineering and Assistant Professor Carson Bruns of the Paul M. Rady Department of Mechanical Engineering and ATLAS Institute received $477,000 from the National Science Foundation to begin this three-year project in January 2021.
“We are investigating an emerging class of materials known as slide ring polymers that resemble beads on a string,” said Vernerey. “When the network is subjected to a controlled stimulus, the bead-like molecules can slide around, which allows for a new way to actuate the material.”
Naturally occurring molecular machines in the body perform vital cell functions, such as gene replication, protein synthesis or transportation of intracellular cargo. Artificial molecular machines—inspired by those in nature—were recognized by the 2016 Nobel Prize, awarded to early pioneers in this area. Now, Bruns and Vernerey aim to scale up these tiny machines from nanoscale to macroscale using networks.
Instead of one molecule, a network incorporates numerous molecules, linked and working together, as occurs naturally in muscle. The process of starting small and scaling up allows the manmade material to copy how nature organizes molecular machines. To ensure the best results, Vernerey will also create a multiscale model to generate predictions that will help determine exactly how to tweak the molecular structure for the most effective scaling.
“The part that Franck’s group is doing is the first of its kind for these materials,” said Bruns. “It keeps my group from having to go into the lab and make hundreds of networked molecular machines until we find the property that we’re most interested in.”
Likewise, Vernerey said there would be no models without Bruns. “We make for a very cool integration,” said Vernerey.
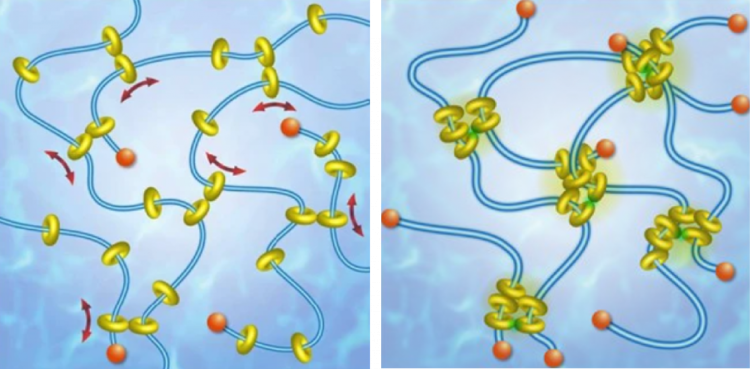
A schematic showing a slide ring network relaxing (left) and contracting (right). The bead-like molecules slide around, allowing for a new way to actuate the material.
Hydrogels currently are the main available actuator based on molecular interactions, which change shape based on temperature, pH or pulses of electricity. However, these actuators are limited in that they cannot change shape in only one dimension. When a soft material experiences a change in volume instead of just length, its movements are slower, harder to control and a less efficient use of energy.
“Imagine the actuator is a sponge soaked in water, and it takes a long time for the water to leave,” said Vernerey. “The larger the actuator, the longer you have to push out. This means when you want to scale it up, this approach becomes unrealistically slow. The only way to make things fast is to contract without volume change.”
Natural muscle, the inspiration for this project, does this quickly in the body. Each molecular machine pulls on polymer ropes in a microscopic tug-of-war, and the movements collectively result in the muscle shortening to contract and fully extending to relax.
“Another consideration is that our materials can be made to be self-healing, and they are biodegradable, both properties of muscle,” said Bruns.
Bruns said their materials are made of non-toxic, food-grade products. This is significant, because most other actuators—especially those that rely on electricity—are not safe to use inside the body.
“While we hope there will be applications, we are equally interested in better understanding these systems,” said Bruns. To this end, Bruns and Vernerey are also developing interactive lessons in this area for high-school and undergraduate students.
“If you tell a student in high school, I’m just building a polymer, they might not be that excited,” said Vernerey. “But this project has great applications which will help them to be excited about the physics.”
Whether their findings help in tissue engineering or in developing soft micro-robots to mimic and guide cells, among other applications, Bruns and Vernerey said they are excited to be on the frontier of nanotechnology, gaining a better understanding of molecular machines and networks.

