How plankton hold secrets to preventing pandemics
Whether it’s plankton exposed to parasites or people exposed to pathogens, a host’s initial immune response plays an integral role in determining whether infection occurs and to what degree it spreads within a population, new CU Boulder research suggests.
The findings, published May 13 in The American Naturalist, provide valuable insight for understanding and preventing the transmission of disease within and between animal species. From parasitic flatworms transmitted by snails into humans in developing nations, to zoonotic spillover events from mammals and insects to humans—which have caused global pandemics like COVID-19 and West Nile virus—an infected creature’s immune response is a vital variable to consider in calculating what happens next.
“One of the biggest patterns that we’re seeing in disease ecology and epidemiology is the fact that not all hosts are equal,” said Tara Stewart Merrill, lead author of the paper and a postdoctoral fellow in ecology. “In infectious disease research, we want to build host immunity into our understanding of how disease spreads.”

Tara Stewart Merrill. (Credit: Loren Merrill)
Invertebrates are common vectors for disease, which means they can transmit infectious pathogens between humans or from animals to humans. Vector-borne diseases, like malaria, account for almost 20% of all infectious diseases worldwide and are responsible for more than 700,000 deaths each year.
Yet epidemiological studies have rarely considered invertebrate immunity and recovery in creatures that are vectors for human disease. They assume that once exposed to a pathogen, the invertebrate host will become infected.
But what if it was possible for invertebrates to fight off these diseases, and break the link in the chain that passes them on to humans?
While observing a tiny species of zooplankton (Daphnia dentifera) throughout its lifecycle and exposure to a fungal parasite (Metschnikowia bicuspidata), the researchers saw this potential in action. Some of the plankton were good at stopping fungal spores from entering their bodies, and others cleared the infection within a limited window of time after ingesting the spores.
“Our results show that there are several defenses that invertebrates can use to reduce the likelihood of infection, and that we really need to understand those immune defenses to understand infection patterns,” said Stewart Merrill.
Unexpected recovery
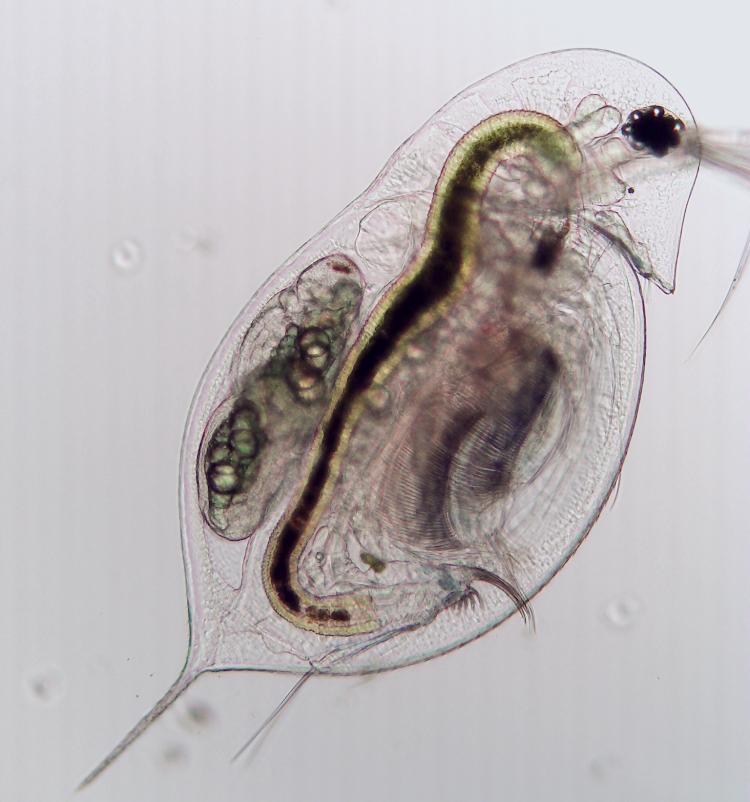

Daphnia. (Credit: Tara Stewart Merrill)
Stewart Merrill started this work in her first year as a doctoral student at the University of Illinois, studying this little plankton and its collection of defenses. It’s a gruesome process if the plankton fails to ward off the parasite: Its fungal spores attack the plankton’s gut, fill its body and grow until they are released when the host finally dies.
But she noticed something that had not been recorded before: Some of the doomed plankton recovered. Several years later, she has found that when faced with identical levels of exposure, the success or failure of these infections depends on the strength of the host’s internal defenses during this early limited window of opportunity.
Based on their observations of these individual outcomes, the researchers developed a simple probabilistic model for measuring host immunity that can be applied across wildlife systems, with important applications for diseases transmitted to humans by invertebrates.
“When immune responses are good, they act as a filter that reduces transmission,” said Stewart Merrill. “But any environmental change that degrades immunity can actually amplify transmission, because it will let all of that exposure go through and ultimately become infectious.”
It’s a model that can also apply to COVID-19, as research from CU Boulder has shown that not all hosts are the same in transmitting the coronavirus, and exposure does not directly determine infection.
COVID-19 is also believed to be the result of a zoonotic spillover, an infection that moved from animals into people, and similar probabilistic models could be advantageous in predicting the occurrence and spread of future spillover events, said Stewart Merrill.
Understanding prevention of infection
Stewart Merrill hopes that a better understanding of infections in a simple animal like plankton can be applied more broadly to invertebrates that matter for human health.
In Africa, Southeast Asia, as well as South and Central America, 200 million people suffer from infections caused by schistosomes—invertebrates more commonly known as parasitic flatworms. They cause illness and death, and significant economic and public health consequences, so much so that the World Health Organization considers them the second-most socioeconomically devastating parasitic disease after malaria.
They’re just one of many neglected tropical diseases transmitted to people by invertebrate hosts such as snails, mosquitoes and biting flies. These diseases infect a large portion of a population but occur in areas with low levels of sanitation that don't have the economic resources to address those diseases, said Stewart Merrill.
Schistosomes live in freshwater environments that people use for their drinking water, laundry and bathing. So even though there are treatments, the next day a person can easily get reinfected just by accessing the water they need. By better understanding how the flatworms themselves succumb to or fight off infection, scientists like Stewart Merrill help us get closer to stopping the chain of transmission into humans.
“We really need to work on understanding prevention of infection, and what that risk is in those aquatic systems, rather than just cures for infection,” she said.
The good news is we can learn from the same invertebrates which infect us. In invertebrate hosts that suffer or die from their infections, there is a good incentive to learn how to build an immune response and fight it off. Some snails have even shown the ability to retain an immunological memory: If they get infected once and survive, then they might never get infected again.
“If we can better understand how the environment shapes those defenses, we could predict into the future how environmental changes might amplify or suppress risk of transmission to people,” said Stewart Merrill.
Additional authors on this paper include Zoi Rapti and Carla Cáceres at the University of Illinois.


