Mimicking the heart’s microenvironment
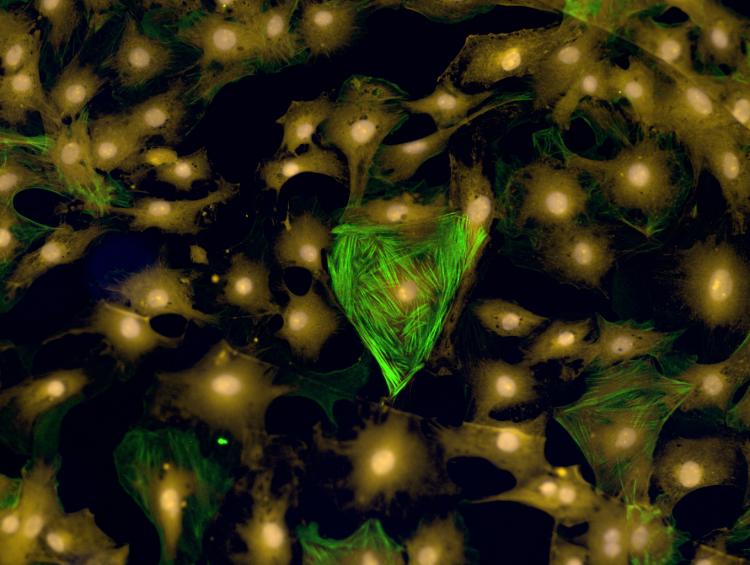
Rat cardiac fibroblasts—which happen to be in the shape of a heart—grown on hydrogels mimicking cardiac tissue and treated with human serum. (Photo: Brian Aguado)
CU Boulder engineers and faculty from the Consortium for Fibrosis Research & Translation (CFReT) at the CU Anschutz Medical Campus have teamed up to develop biomaterial-based “mimics” of heart tissues to measure patients’ responses to an aortic valve replacement procedure, offering new insight into the ways that cardiac tissue re-shapes itself post-surgery.
Aortic valve stenosis (AVS), a progressive disease characterized by heart valve tissue stiffening and obstructed blood flow from the heart, is known as a “silent killer,” affecting 12.4% of the population over 75 years old with a mortality range of 2-5 years if left untreated. Transcatheter aortic valve replacement (TAVR) procedures, which place an artificial valve at the site of the blockage, have been widely and successfully adopted as a remedy in recent decades.
Details of the broader biological reaction to the valve replacement have remained largely unknown, but nevertheless hold significant ramifications for quantifying the quality of recovery, the risk of complications and the assessment of overall patient outcomes.
During AVS disease progression, tissue-specific cells known as fibroblasts transition into myofibroblasts, which promote tissue stiffening. The researchers were interested in understanding how and why, post TAVR, myofibroblasts revert to the more benign fibroblasts.
“Previous studies have shown significant remodeling of cardiac tissues post-intervention,” said Dr. Brian Aguado, lead author of the study and a post-doctoral researcher in CU Boulder’s Department of Chemical and Biological Engineering. “Our hypothesis was that perhaps there are biochemical cues in a patient’s blood that may revert myofibroblasts back to fibroblasts.”
Modeling such a transformation in the lab is one thing, Aguado said, but the key to the new study was obtaining blood samples from real AVS patients and then using biomaterials to replicate the microenvironment of the heart.
“The heart is not made of plastic like a petri dish is,” he said. “We needed to engineer materials that could reflect the various stiffnesses of both healthy and diseased valve and cardiac tissue.”
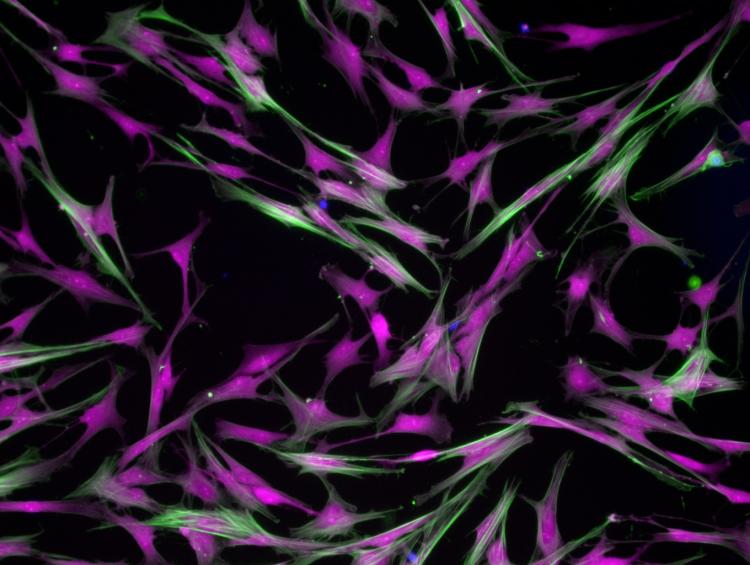
Porcine valve fibroblasts grown on hydrogels mimicking valve tissue and treated with human serum. (Photo: Brian Aguado)
The researchers collected blood serum samples from AVS patients both pre- and post-TAVR procedure and then treated cardiac cells cultured in a customized hydrogel environment, maintaining a near-facsimile of the in vivo cardiac conditions before and after TAVR.
The researchers were able to quantify protein expression in patient sera, identifying key proteins associated with myofibroblast deactivation as the aortic and cardiac tissue re-shapes and rebuilds itself after TAVR.
"Our lab is focused on engineering hydrogels as mimics of the extracellular tissue microenvironment," said Dr. Kristi Anseth, distinguished professor of chemical and biological engineering and director of the Precision Biomaterials IRT. “The hydrogel system developed for these studies enable us to evaluate how patient-specific biochemical cues, found in human sera, can impact cellular phenotypes. Our patient-specific observations would not have been possible using conventional tissue culture plastic materials."
“Cardiac fibrosis due to excess deposition of extracellular matrix proteins is a massive problem,” said Timothy McKinsey, professor of medicine and director of the CFReT, one of the programs supported through the University of Colorado School of Medicine's Transformational Research Funding initiative. “Among other things, fibrosis causes the heart to become stiff, impairing its ability to relax. We are excited about the potential of translating our current findings to develop innovative therapies for fibrotic diseases of the heart and vasculature.”
The research may also yield future insight into the observed differences in recovery between men and women. Previous clinical studies have suggested that men seem to undergo more cardiac tissue remodeling post-TAVR, and the new data found that male cardiac cells do indeed see more pronounced myofibroblast reversal relative to female cells, though further research is needed to understand sex-specific differences in various clinical contexts.
“We were a bit surprised by the breadth of these findings,” Aguado said. “We didn’t think that a valve implant could have such a profound impact on the body system-wide. The connections between our engineered models and clinical data give strength to that conclusion.”
Overall, Aguado said, the results show that TAVR procedures do indeed trigger a beneficial protein response and that biomaterial models together with clinical samples can provide a useful bridge toward identifying future therapeutic opportunities.
“We are getting better at engineering disease models, but we’re reaching a crossroads where models can only do so much,” he said. “The future will rely on using patient samples in conjunction with these models to better understand disease progression in a patient. In collaboration with physicians, we can see how our advances in the lab can be translated into identifying more effective treatments for patients.”
The new study was published today in the journal Science Translational Medicine. Additional co-authors of the new study include Joseph Grim, Cierra Walker, Tova Ceccato, Anne Cox and Leslie Leinwand of CU Boulder; Katherine Schuetze and Carmen Sucharov of the CU Anschutz Medical Campus; Aik-Choon Tan of the Colorado School of Public Health; and Matthew Taylor of the University of Colorado Health Science Center. The National Institutes of Health (NIH), the National Science Foundation (NSF), the American Heart Association, the U.S. Department of Education and the Burroughs Wellcome Fund provided funding for the research.


