Nanostructured Polymer Electrolytes for Enhanced Ion Conductivity (collaborative work with Dr. Brian Elliott, TDA Research, Inc.)
Very recently, a new lyotropic LC monomer with a lithium sulfonate headgroup (7) has been prepared that forms a type II bicontinuous cubic (QII) phase in the presence of propylene carbonate (PC) and Li salt solutions of PC, instead of water (Figure 9). After cross-linking of the QII phase, AC impedance studies show that a nanochannelled, lyotropic LC composite of 7 containing 15 wt % (0.245 M LiClO4 in PC) exhibits a room-temperature ion conductivity in the low- to mid 10–3 S/cm range. This value is comparable to the Li ion conductivity values observed for conventional 1.0 M Li salt-doped, liquid electrolyte-gelled poly(ethylene oxide)-based Li ion battery electrolytes. In addition, this new lyotropic LC solid-liquid nanocomposite is able to retain an ion conductivity of ca. 10–4 S/cm down to –35 °C, a temperature at which conventional solid-state and gelled PEO-based polymer electrolytes are effectively immobile and poorly conducting (<10–7 S/cm). It is also able to retain an ion conductivity of ca. 10–4 S/cm when heated up to 100 °C. This new type of liquid-filled, nanostructured polymer electrolyte would be valuable in the design of better Li ion batteries and related electronic devices that can operate over a wider temperature range. The fact that the QII-phase polymer nanocomposite is able to afford “liquid-like” ion conductivity over a wide temperature range in a stable, flexible solid film material is unique. Preliminary work with this material in making working prototype Li-metal coin cell batteries has been successful, and shows the promise of this new material in battery fabrication.
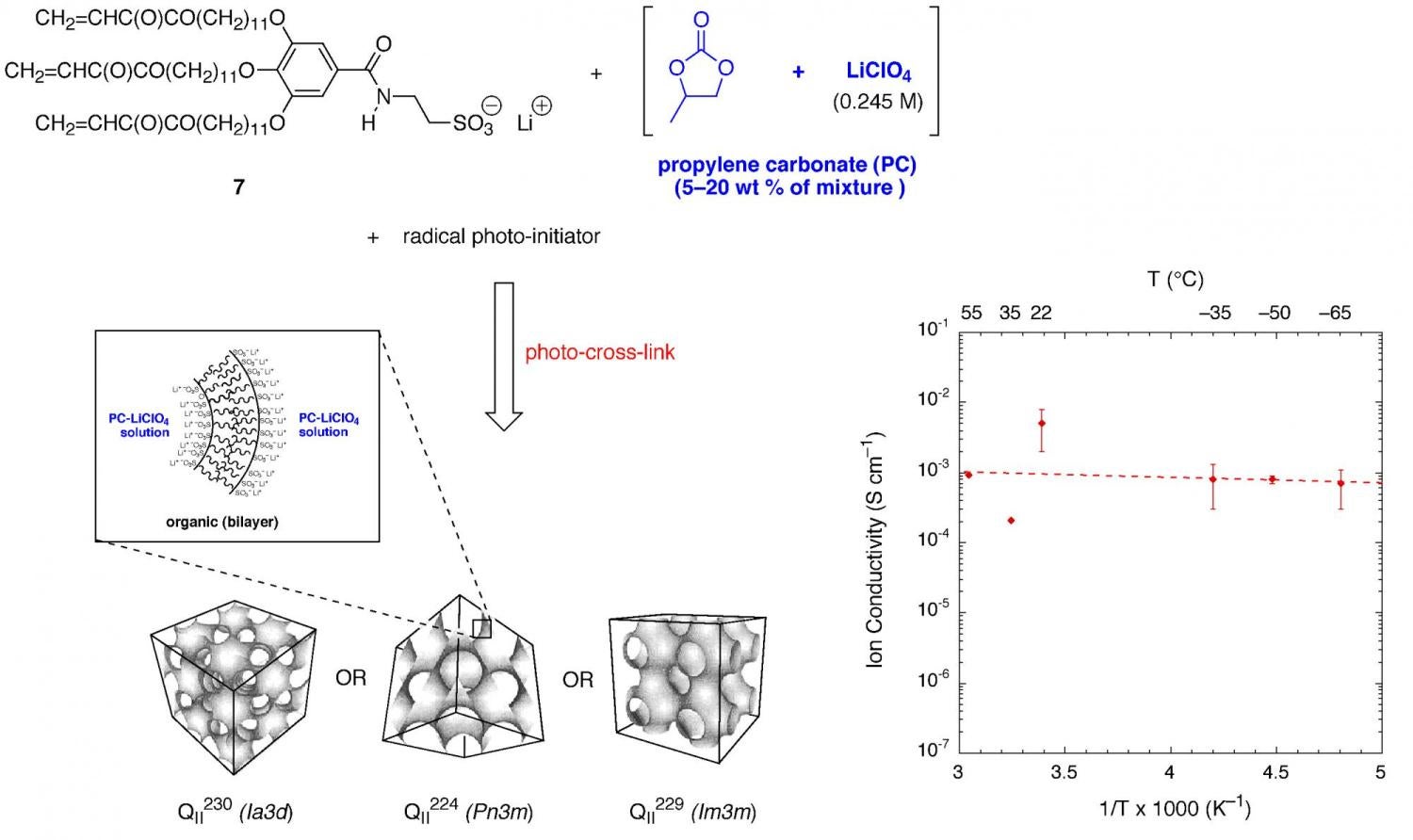
Figure 9. Cross-linked QII phases of monomer 7 formed with LiClO4-PC solutions as a new type of polymer electrolyte material for Li batteries with stable ion conductivity over a wide temperature range.

