Design and Investigation of New Types of Functionalized RTILs for CO2 and Toxic Vapor Separations
Typically, RTILs are organic salts based on ammonium, phosphonium, pyridinium, or most commonly imidazolium cations, plus a large relatively non-Lewis basic anion, such as a large halide, BF4–, PF6–, bis(trifluoromethanesulfonimide)–, etc. that prevents crystallization. Typically, RTIL cations are functionalized with alkyl substituents to give stable quaternary N- or P-based cationic centers needed in the organic cation. Although relatively easy to synthesize, alkyl functionalized RTIL cations do not impart additional functional properties outside of control of overall hydrophobic vs. hydrophilic character. Our group has focused on synthesizing new RTIL derivatives based on imidazolium cations containing new functional units other than conventional alkyl substituents, in order to impart new physical or chemical properties to RTILs. For example, we have prepared new imidazolium-based RTILs containing oligo(ethylene oxide), alkylnitrile, fluoroalkyl, polyalcohol, and oligo(siloxane) substituents on the cation (Figure 15), in order to improve the solubility of CO2 and other targeted light gases over other gases in gas separations. In the course of this work, we can also show that classical group contribution theory for rationalizing solubilities of materials in other materials, can be applied to the design of substituted RTILs. We are in the process of designing and synthesizing even more advanced functionalized RTILs with specific reactive and/or catalytic properties.
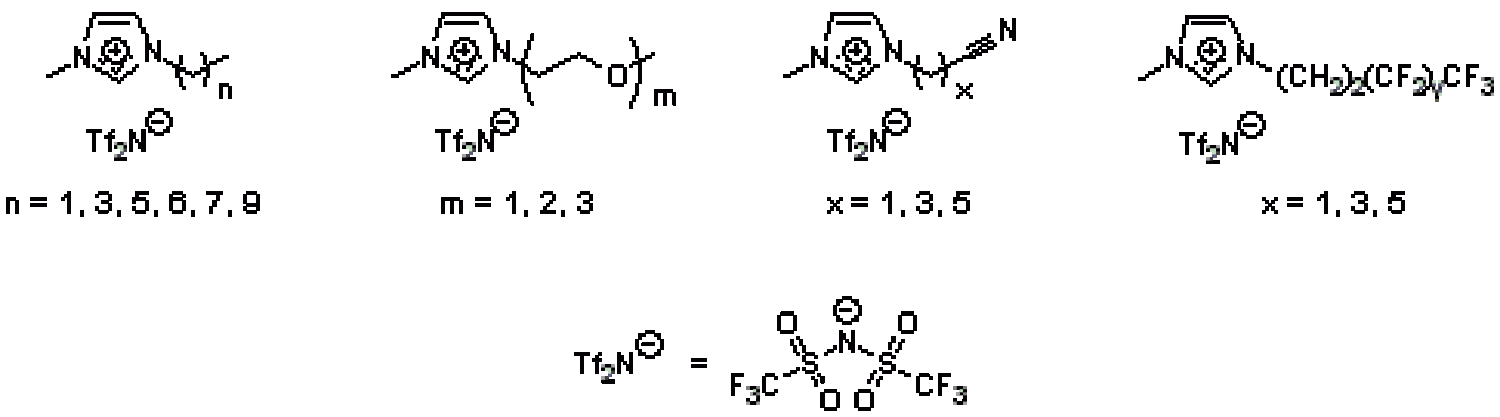
Figure 15. Some of the functionalized imidazolium-based RTILs prepared by our group and studied for CO2 separation and uptake from other light gases.
We are also in the process of designing and synthesizing even more advanced, functionalized RTILs with specific reactive and/or catalytic groups for CWA and TIC capture. One example is a Lewis-acidic RTIL containing a pendant borane (i.e., 9-borobicyclononane (9-BBN)) group that can dative bond with Lewis-basic TICs, such as ammonia gas, volatile amines, and phosphine compounds (Figure 16). The non-volatile and fluid nature of the RTIL moiety allows the resulting adduct to be potentially more easily handled and contained. Preliminary studies on this RTIL have shown that it will rapidly bind with triethylamine upon liquid contact to form solid, viscous adduct.
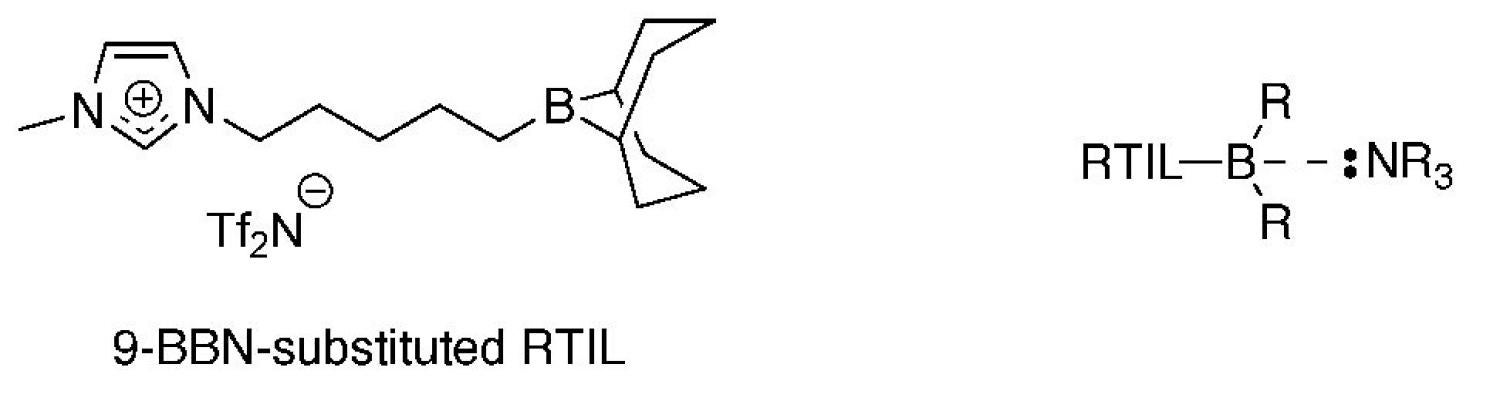
Figure 16. Structures of a new borane-substituted RTIL for hazardous chemical vapor capture of Lewis-basic TICs.

