Stimulated emission depletion (STED) microscopy with polarization-maintaining fiber
Background
STED microscopy is an important technique that can overcome the diffraction limit and resolve structures on the order of ~40mm. It is a powerful method that is particularly useful in a biological setting with fluorophores, where it can be used to monitor subcellular activity in situ. The applicability of STED can be enhanced by moving from free space to fiber optics, opening up the possibility for endoscopic and in vivo sub-diffraction limited imaging. Standard commercially available step index fiber does not support orbital angular momentum modes as eigenmodes and there is therefore a need for improved methods and systems for STED microscopy.
Technology
Researchers at the University of Colorado have come up with a method for STED microscopy of a fluorescent species in an object to be imaged, the fluorescent excitation wavelength, a fluorescence depletion wavelength and a fluorescence emission wavelength.
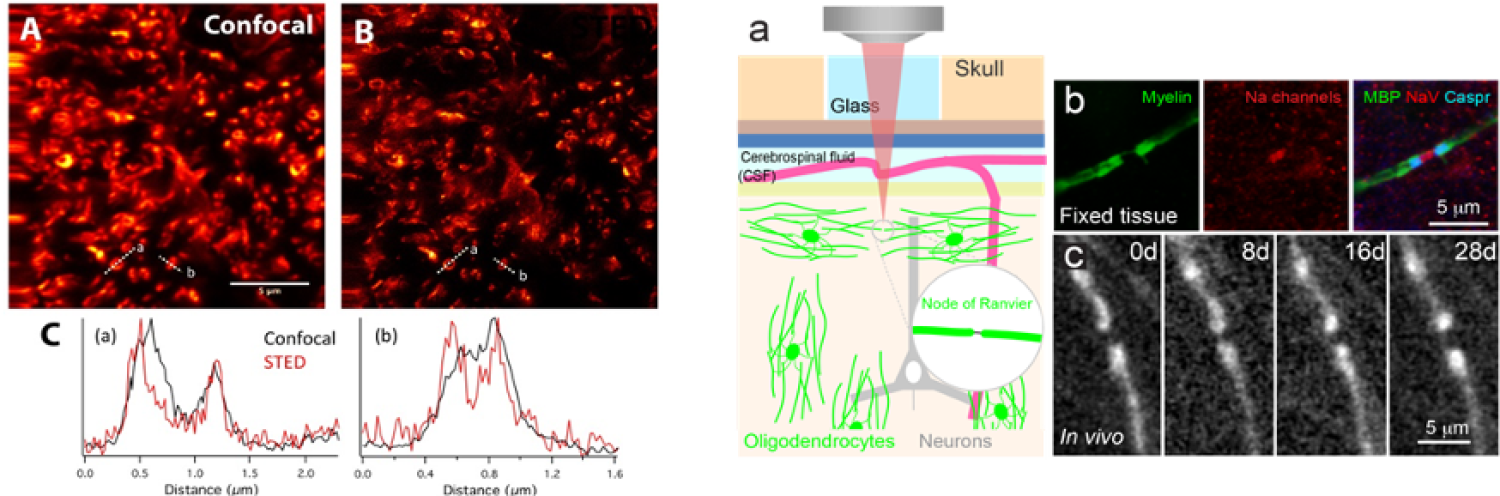
Figure: (Left) Confocal (A) and STED (B) images of oligodendrocytes expressing EGFP in the spinal cord of the Plp-EGFP mouse. Line scan of the intensity measured in places where myelin ensheaths an axon. (Right) In vivo imaging of nodes of Ranvier. a) Diagram demonstrating in vivo two-photon imaging through a cranial window in a Mobp-EGFP mouse. b) Horizontal section from somatosensory cortex of Mobp-EGFP mice immunostained for EGFP, Caspr (paranode marker), and sodium channels (NaV, nodes of Ranvier). c) In vivo imaging of an individual node of Ranvier over 28 days. Note that higher resolution is needed to accurately assess node of Ranvier size.
Advantages
- Ability to study humans and animals on move using STED microscopy providing a more natural environment for them
- Ability to do a STED microscopy examination of patients acting more naturally then when being at a fixed spot
- More comfortable STED microscopy examinations for the patient
Applications
- Medical examination
- Animal studies
- Human and certain drug studies
What's Next?
This technology is available for exclusive or non-exclusive licensing.
Nicole Forsberg: nicole.forsberg@colorado.edu
The Newsroom
For marketing and communication inquiries or news tips, contact Daniel Leonard, senior marketing and communications specialist for Venture Partners at CU Boulder.
For media inquiries, please visit colorado.edu/news/formedia.


