An E1–E2 fusion protein primes antiviral immune signalling in bacteria
Nature (2023) PubMed PMID: 36755092; PubMed Central PMCID: PMC10292035
Abstract
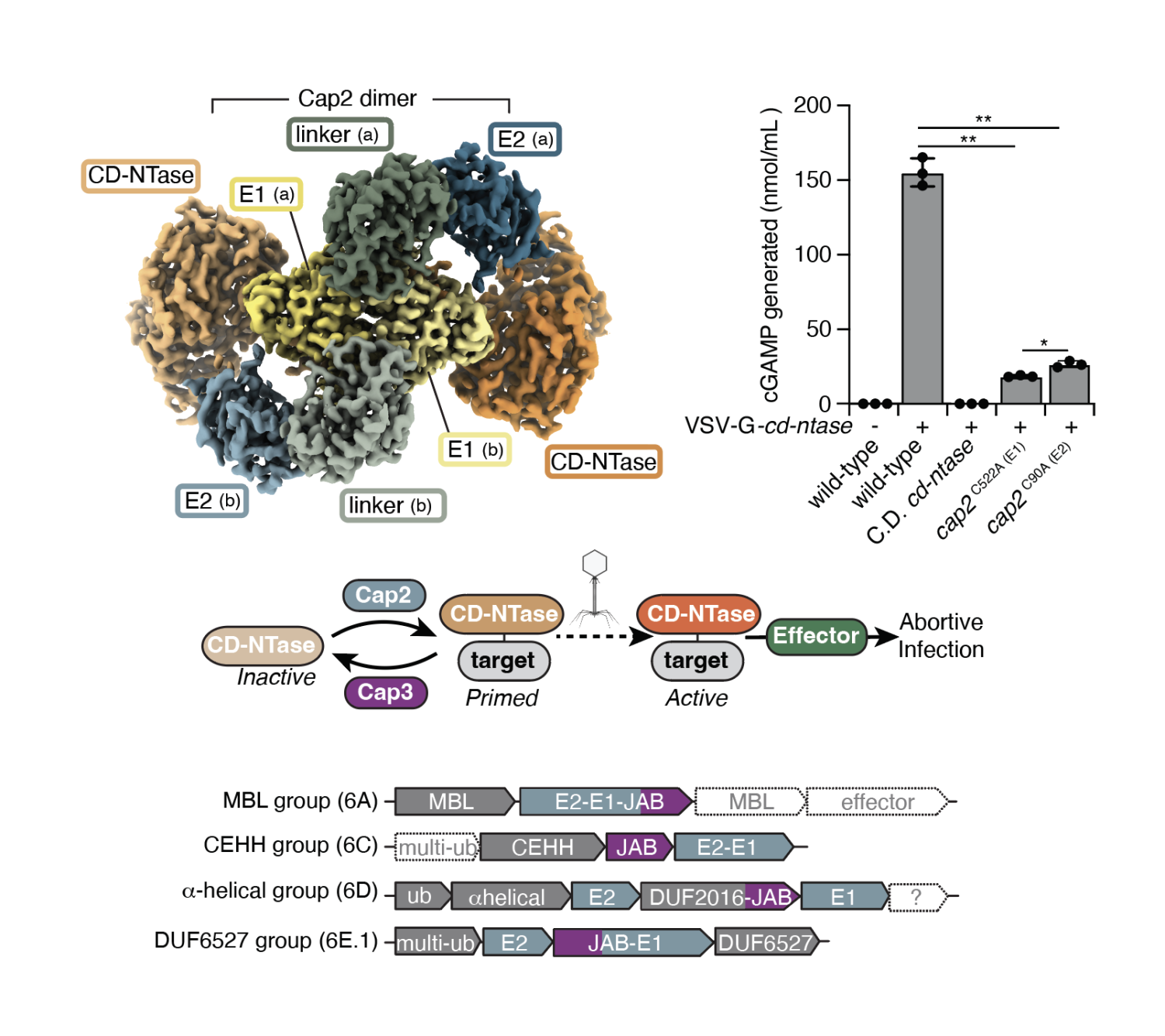
In all organisms, innate immune pathways sense infection and rapidly activate potent immune responses while avoiding inappropriate activation (autoimmunity). In humans, the innate immune receptor cyclic GMP–AMP synthase (cGAS) detects viral infection to produce the nucleotide second messenger cyclic GMP–AMP (cGAMP), which initiates stimulator of interferon genes (STING)-dependent antiviral signalling. Bacteria encode evolutionary predecessors of cGAS called cGAS/DncV-like nucleotidyltransferases (CD-NTases), which detect bacteriophage infection and produce diverse nucleotide second messengers. How bacterial CD-NTase activation is controlled remains unknown. Here we show that CD-NTase-associated protein 2 (Cap2) primes bacterial CD-NTases for activation through a ubiquitin transferase-like mechanism. A cryo-electron microscopy structure of the Cap2–CD-NTase complex reveals Cap2 as an all-in-one ubiquitin transferase-like protein, with distinct domains resembling eukaryotic E1 and E2 proteins. The structure captures a reactive-intermediate state with the CD-NTase C terminus positioned in the Cap2 E1 active site and conjugated to AMP. Cap2 conjugates the CD-NTase C terminus to a target molecule that primes the CD-NTase for increased cGAMP production. We further demonstrate that a specific endopeptidase, Cap3, balances Cap2 activity by cleaving CD-NTase–target conjugates. Our data demonstrate that bacteria control immune signalling using an ancient, minimized ubiquitin transferase-like system and provide insight into the evolution of the E1 and E2 machinery across domains of life.
News and Commentaries
- Scientists discover more bacterial tools that could be reprogrammed to treat human disease [CU Boulder Today]
- Read Hannah's Tweetorial [Twitter]
Links
- DOI: https://doi.org/10.1038/s41586-022-05647-4
- Journal Link: https://www.nature.com/articles/s41586-022-05647-4
- BioRxiv Preprint, March 31 2022, https://www.biorxiv.org/content/10.1101/2022.03.31.486616
Citation
Ledvina HE, Ye Q, Gu Y, Sullivan AE, Quan Y, Lau RK, Zhou H, Corbett KD, Whiteley AT. An E1-E2 fusion protein primes antiviral immune signalling in bacteria. Nature. 2023 Apr;616(7956):319-325. doi: 10.1038/s41586-022-05647-4. Epub 2023 Feb 8. PubMed PMID: 36755092; PubMed Central PMCID: PMC10292035.