Ella Hushka
- Graduate Student
- CHEMICAL AND BIOLOGICAL ENGINEERING
Curriculum Vitae
Research Interests
Intestinal organoids hold unparalleled biomimicry and consequently great promise for studying organogenesis, modeling disease and screening drug candidates in vitro. To date, intestinal organoids are largely cultured in Matrigel, a protein rich biological matrix with an ill-defined composition and limited user control of properties. As a result, there has been growing interest in development of synthetic hydrogel systems with tailored properties for intestinal organoid culture, enabling opportunities to better understand the role of biochemical and mechanical matrix cues on intestinal development and evolution of disease. To that end, Ella employs a suite of poly(ethylene glycol) (PEG) hydrogels with dynamic crosslinkers that allow precise tuning of the relaxation of forces and formation of differentiated organoids. By culturing ISCs in these materials, Ella tests hypotheses related to the influence of matrix force relaxation on crypt forming efficiency, length and number, matricellular signaling required for intestinal differentiation and correlations between matrix mechanics and crypt cellular structure, organization and function.
Outside of lab Ella enjoys biking, calligraphy and advocating for other disabled scientists.
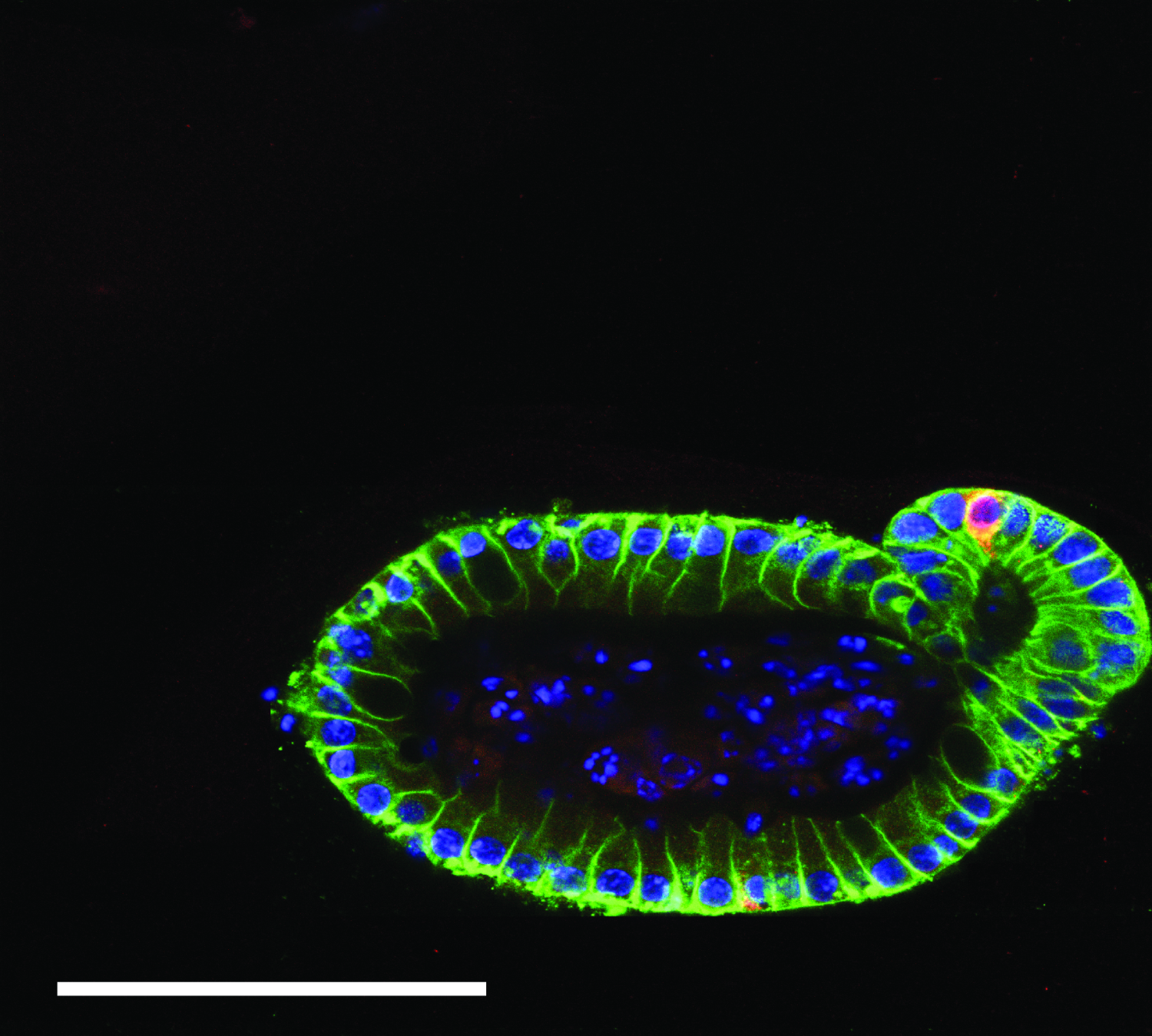
Figure 1. Intestinal organoid culture in photodegradable hydrogels, immunostained for nuclei (DAPI, blue), e-cadherin (green) and enteroendrocrine cells (Chromogranin A, red).


