Amie (Tze-Ling) Chang
- Postdoctoral Researcher
- CHEMICAL AND BIOLOGICAL ENGINEERING

Curriculum Vitae
Research Interests
Skeletal muscle is a highly specialized tissue and has a remarkable capability of regeneration after injury. However, the ability to repair and renew in aged muscle tissue is significantly diminished compared to healthy adult tissue. Previous studies have shown that muscle ECM undergoes remodeling after injury, and this remodeling plays a significant role in regulating regeneration by directing the fate of muscle satellite cells (MuSCs). For example, activated MuSCs remodel their niche with increased fibronectin, which stimulates symmetric expansion of MuSCs to maintain the stem cell pool during regeneration. However, other studies have shown that there is a reduced level of fibronectin in aged satellite cell niche. In addition, it has been demonstrated that aged MuSCs have less integrin activity and lower YAP/TAZ expression. However, how MuSCs are affected by the ECM proteins in the niche and how this interaction is influenced during aging still remain unclear. My research aims to investigate how muscle satellite cells (MuSCs) on a myofiber interact with the extracellular matrix and how aging influences cell-matrix interactions. In order to achieve this goal, myofibers were encapsulated in synthetic hydrogels with tunable ECM peptide compositions. Also, a photo-expansion microscopy technique was applied to better visualize the YAP/TAZ translocation and the cell-matrix interaction. We hope that this study can help us understand how the satellite niche affects satellite cell behaviors, and thus shedding light on how we might be able to do to help maintain the function of satellite cells and facilitate aged muscle regeneration.
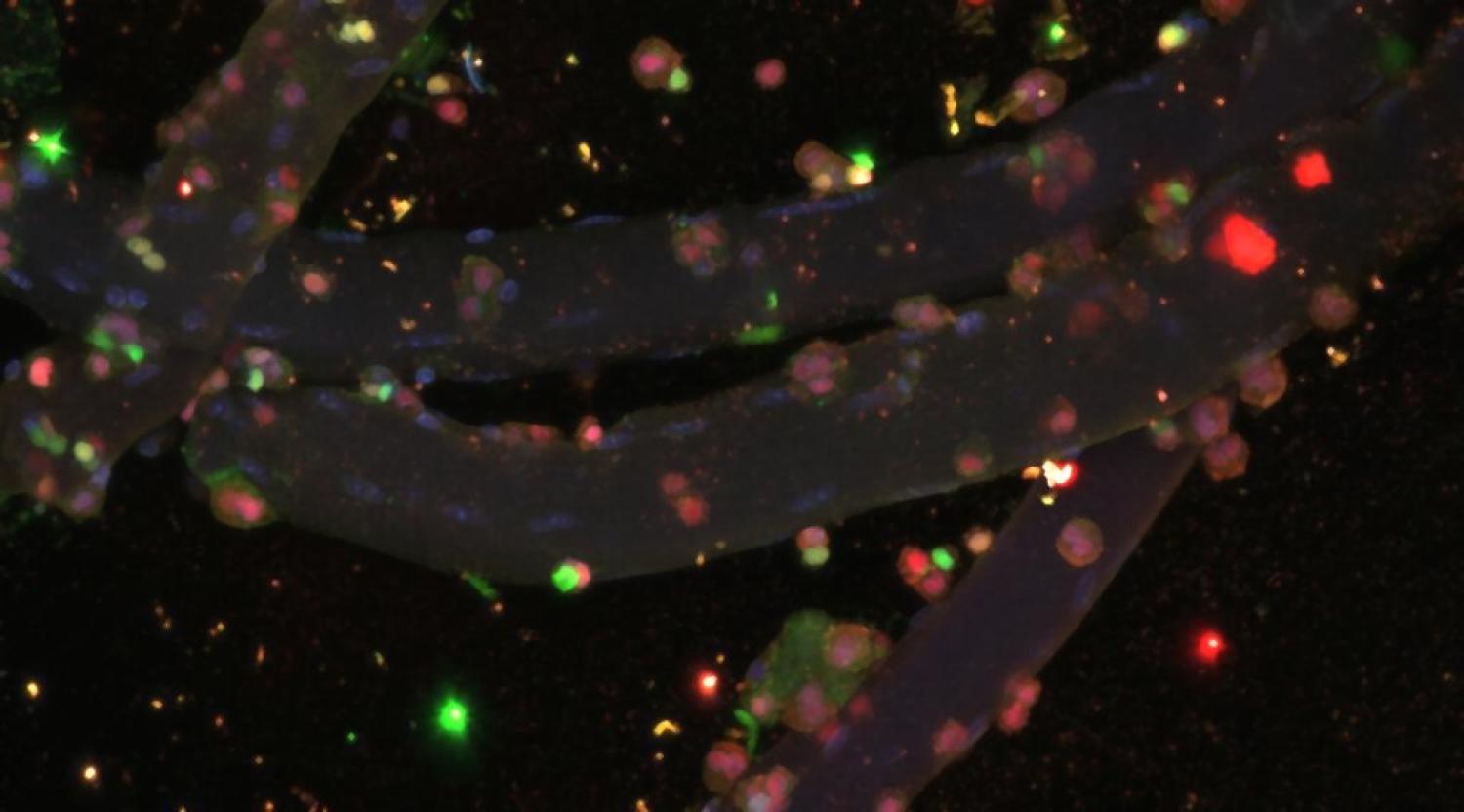
Encapsulated Myofibers. Blue: DAPI, Green: Pax7, Pink: MyoD, Red: Myogenin

